Why Europeans Are So White-Skinned
… and other possible outcomes of sexual selection in European populations
by Peter Frost
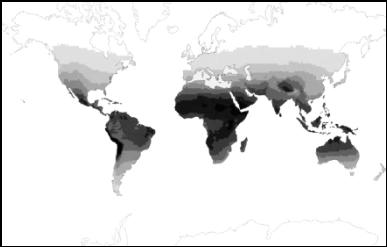
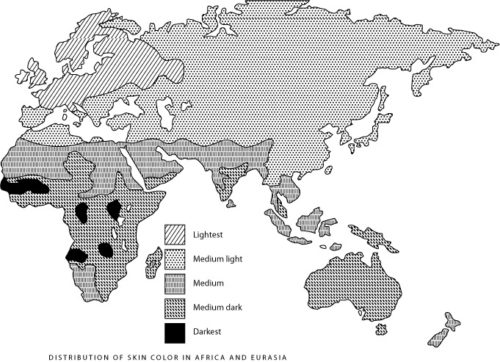
DID SEXUAL SELECTION alter the appearance of Europeans in a number of ways, and not simply by diversifying hair and eye color? (ILLUSTRATION: Figure 1. Distribution of skin color in Africa and Eurasia (Brace, 1973))
It may have accentuated existing sexual dimorphisms. Several studies have found wider hips, narrower waists, and thicker deposition of subcutaneous fat in women of European descent than in other women (Hrdlicka, 1898; Meredith & Spurgeon, 1980; Nelson & Nelson, 1986). This accentuation is noticeable even before birth: Euro-American fetuses are sexually more dimorphic than African-American fetuses (Choi & Trotter, 1970).
The proximal cause seems to be lower androgen production than in women of sub-Saharan African descent (Falkner et al., 1999) and higher estrogen production and lower fecal excretion of estrogen than in women of north/east Asian descent (Adlercreutz et al., 1994; Coker et al., 1997; Key et al., 1990; Taioli et al., 1996; Wang et al., 1991). Prenatal exposure to estrogen, as indicated by “digit ratio” (ratio of second finger length to fourth finger length), also seems higher in European women, albeit with much inter-population variation (Manning et al., 2000; Manning, 2003). This variation may reflect a maternal-age effect: digit ratio is higher in Catholic countries like Poland and Spain, where mothers generally bear children in their twenties, than in Germany and Finland, where they tend to wait until their thirties (Manning et al., 2000).
Sexual selection may have also lightened European skin. Consider the extreme depigmentation of northern and eastern Europeans. Their white skin deviates markedly from the much weaker north-south gradient in skin color of other human populations (the latter gradient may reflect selection to maintain sufficient vitamin D synthesis, see Figure 1). Although their geographical area does not coincide with a zone of less intense exposure to UV radiation (Figure 2), it does coincide with the geographical area of hair and eye color diversification. Aside from red hair and blue eyes, these color traits show little genetic linkage to skin color (Duffy et al. 2004; Flanagan et al. 2000; Sturm et al. 2001; Sturm and Frudakis 2004). It looks as though a common selective force acted simultaneously on skin, hair and eye color within this geographic area while not acting on populations living at the same latitudes in northern Asia and North America (Frost, 1994a; Manning et al., 2004).
If this selective force is sexual selection, it may have lightened European skin by acting on an existing sexual dimorphism. Complexion differs between the sexes because of differing amounts of melanin and cutaneous blood flow; in short, women are fairer; men browner and ruddier (Edwards & Duntley, 1939; Frost, 1988; Frost, 2005; Hulse, 1967; Jablonski & Chaplin, 2000).
The size of this sex difference is still unsure. First, most studies are poorly controlled for age, yet girls lighten only after puberty and immediately before are actually darker than boys. Second, investigators try to limit tanning by measuring under the arm, yet subcutaneous fat is thinner there and skin pigmentation probably less dimorphic, given the correlation in women between lightness of skin color and thickness of subcutaneous fat (Mazess, 1967). Female skin is probably at its palest in women of childbearing age and on highly adipose parts of the body, i.e., the breasts, the hips, the buttocks, and the thighs.
In any event, this mark of feminity may have been targeted by sexual selection, as suggested by a cross-cultural male preference for lighter-complexioned women (Aoki, 2002; Feinman & Gill, 1978; Frost, 1988; Frost, 1994b; Frost 2005; van den Berghe & Frost, 1986). This selection is apparent in the tendency of upper classes throughout the world to appropriate the fairest-skinned women available. In India:
Wealthy landowning families often have a tradition of seeking light-skinned brides among poorer members of their subcaste. It is very common to find a high concentration of lighter-skinned people among established land-owning families (Béteille 1967).
This selection is also apparent in Japan. In a study of untanned skin, Hulse (1967) found less pigmentation in upper-class Japanese than in lower-class Japanese, even when the latter worked in factories and not on farms. The likeliest explanation is that upper class men have more latitude in choosing wives and tend to take the fairest-skinned ones, thus causing their social class to lighten in color with each passing generation. Hulse notes that “skin color has long been regarded, by the Japanese, as one of the criteria for evaluating physical attractiveness, especially in young females.”
Even if this selection pressure had acted only on ancestral European women, both sexes would have lightened in color. This is because most skin-color genes are not sex-linked. Nonetheless, some are, so women should have lightened a bit more than men did, thus making skin color more dimorphic in Europeans than in other populations. Yet the reverse seems to be true (Frost, in press; Madrigal and Kelly, 2006). The reason may be a ceiling effect, i.e., as ancestral Europeans approached the limit of maximum skin depigmentation, this lightening would have become increasingly hard to achieve for women — already lighter-skinned — than for men. Even though sexual selection had operated primarily on women, men would have lightened more.
How can we test this sexual selection hypothesis? There are two possible lines of approach:
Examine the alleles that depigmented Europeans for possible sex-linkage
According to the sexual selection hypothesis, if some alleles lighten female skin more than male skin, they should be more frequent in Europeans than in other populations, even if this sex-linkage is less effectively expressed in fair-skinned individuals.
We can test this prediction by examining alleles that lighten skin color, are specific to European populations, and appear to have been under positive selection, i.e., the “redhead” MC1R alleles, the derived SLC24A5, SLC45A2, and AIM1 (MATP) alleles, and possibly DTNBP1, EDA, OCA2, KITLG, MYO5A, HPS7, and TYRP1 variants (McEvoy et al. 2006; Sturm and Frudakis 2004). If any of these alleles affect one sex more than the other, they should do so in the direction of lightening female skin more than male skin.
The exercise could then be repeated with other skin color alleles from other human populations to see whether alleles that lighten female skin more than male skin have the same incidence and prevalence in these populations as they do in Europeans.
Date the alleles that depigmented Europeans
According to the sexual selection hypothesis, European skin became whiter when Europe was predominantly steppe-tundra 25,000 to 10,000 years ago. If the cause was solely natural selection to promote vitamin D synthesis, or relaxation of selection for protection from sunburn and skin cancer, the depigmentation should have begun as soon as modern humans became established in Europe, i.e., c. 40,000 BP.
To settle this question, we could retrieve DNA from human skeletal remains for the period from 40,000 BP to 10,000 BP, i.e., from the arrival of modern humans in Europe to the end of the ice age. We could then plot the appearance and evolution over time of the depigmentation alleles.
Alternately, we could date this evolutionary event by indirect means, i.e., by calculating how many new mutations have arisen among European depigmentation alleles since they last shared a common ancestor. The second method has been tried for the AIM1 locus, whose European-specific alleles seem to date back only to 11,000 BP (Soejima et al. 2005).
In conclusion, sexual selection may have acted on all three color traits that characterize the populations of northern and eastern Europe, with hair and eye color being diversified and skin color lightened. This hypothesis is consistent with the narrow timeframe for evolution of these traits, their geographic distribution and the large number of alleles involved.
References
Adlercreutz, H., S.L. Gorbach, B.R. Goldin, M.N. Woods, J.T. Dwyer, and E. Hämäläinen (1994). Estrogen metabolism and excretion in Oriental and Caucasian Women. Journal of the National Cancer Institute, 86, 1076-1082.
Aoki, K. (2002). Sexual selection as a cause of human skin colour variation: Darwin’s hypothesis revisited. Annals of Human Biology, 29, 589-608.
Béteille, A. (1967). Race and descent as social categories in India, Daedalus 96, 444-463.
Brace, C.L., (1973). A nonracial approach towards the understanding of human diversity. In (C.L. Brace and J. Metress eds.). Man in Evolutionary Perspective. Wiley.
Choi, S.C. and M.A. Trotter (1970). Statistical study of the multivariate structure and race-sex differences of American White and Negro fetal skeletons. American Journal of Physical Anthropology, 33, 307-312.
Coker, A.L., M.M. Crane, R.P. Sticca, and D.W. Sepkovic (1997). Re: Ethnic differences in estrogen metabolism in healthy women. Journal of the National Cancer Institute, 89, 89-90.
Duffy, D.L., N.F. Box, W. Chen, J.S. Palmer, G.W. Montgomery, M.R. James, N.K. Hayward, N.G. Martin, and R.A. Sturm. 2004. Interactive effects of MC1R and OCA2 on melanoma risk phenotypes. Human Molecular Genetics 13, 447-461.
Edwards, E.A. and S.Q. Duntley (1939). The pigments and color of living human skin. American Journal of Anatomy, 65, 1-33.
Falkner, B., K. Sherif, A. Sumner, and H. Kushner (1999). Hyperinsulinism and sex hormones in young adult African Americans. Metabolism-Clinical and Experimental 48, 107-112.
Feinman, S. and G.W. Gill (1978). Sex differences in physical attractiveness preferences. Journal of Social Psychology,105, 43-52.
Flanagan, N., E. Healy, A. Ray, S. Philips, C. Todd, I.J. Jackson, M.A. Birch-Machin, and J.L. Rees (2000). Pleiotropic effects of the melanocortin 1 receptor (MC1R) gene on human pigmentation. Human Molecular Genetics, 9, 2531-2537.
Frost, P. (in press). “Comment on Human skin-color sexual dimorphism: A test of the sexual selection hypothesis”, American Journal of Physical Anthropology
Frost, P. (2005). Fair Women, Dark Men. The Forgotten Roots of Color Prejudice. Cybereditions: Christchurch (New Zealand).
Frost, P. (1994a). Geographic distribution of human skin colour: A selective compromise between natural selection and sexual selection? Human Evolution, 9, 141-153.
Frost, P. (1994b). Preference for darker faces in photographs at different phases of the menstrual cycle: Preliminary assessment of evidence for a hormonal relationship, Perceptual and Motor Skills, 79, 507-514.
Frost, P. (1988). Human skin color: A possible relationship between its sexual dimorphism and its social perception. Perspectives in Biology and Medicine, 32, 38-58.
Hrdlicka, A. (1898). Physical differences between White and Colored children. American Anthropologist, 11, 347-50.
Hulse, F.S. (1967). Selection for skin color among the Japanese. American Journal of Physical Anthropology, 27, 143-156.
Jablonski, N.G. and G. Chaplin (2000). The evolution of human skin coloration. Journal of Human Evolution, 39, 57-106.
Key, T.J.A., J. Chen, D.Y. Wang, M.C. Pike, and J. Boreham (1990). Sex hormones in women in rural China and in Britain. British Journal of Cancer, 62, 631-636.
Madrigal L and W. Kelly 2006. Human skin-color sexual dimorphism: A test of the sexual selection hypothesis. American Journal of Physical Anthropology (online pub.www.interscience.wiley.com )
Manning, J.T., P.E. Bundred, and F.M. Mather (2004). Second to fourth digit ratio, sexual selection, and skin colour. Evolution and Human Behavior, 25, 38-50.
Manning, J.T. (2003). Personal communication.
Manning, J.T., L. Barley, I. Lewis-Jones, J. Walton, R.L. Trivers, R. Thornhill, D. Singh, P. Rhode, T. Bereckzei, P. Henzi, M. Soler, and A. Sved (2000). The 2nd to 4th digit ratio, sexual dimorphism, population differences and reproductive success: evidence for sexually antagonistic genes. Evolution and Human Behavior, 21,163-183.
Mazess, R.B. (1967). Skin color in Bahamian Negroes. Human Biology, 39, 145-154.
McEvoy, B., S. Beleza, and M.D. Shriver. (2006). The genetic architecture of normal variation in human pigmentation: an evolutionary perspective and model. Human Molecular Genetics, 15, R176-R181.
Meredith, H.V. and J.H. Spurgeon (1980). Somatic comparisons at age 9 years for South Carolina White Girls and girls of other ethnic groups. Human Biology, 52, 401-411.
Nelson, J.K. and K.R. Nelson (1986). Skinfold profiles of Black and White boys and girls ages 11-13. Human Biology, 58, 379-390.
Soejima, M., H. Tachida, T. Ishida, A. Sano, and Y. Koda (2005). Evidence for recent positive selection at the human AIM1 locus in a European population. Molecular Biology and Evolution, 23, 179-188.
Sturm, R.A. and T.N. Frudakis (2004). Eye colour: portals into pigmentation genes and ancestry. Trends in Genetics 20, 327-332.
Sturm, R.A., R.D. Teasdale, and N.F. Box (2001). Human pigmentation genes: identification, structure and consequences of polymorphic variation. Gene, 277, 49-62.
Taioli, E., S.J. Garte, J. Trachman, S. Garbers, D.W. Sepkovic, M.P. Osborne, S. Mehl, and H.L. Bradlow (1996). Ethnic differences in estrogen metabolism in healthy women. Journal of the National Cancer Institute, 88, 617.
Van den Berghe, P.L. and P. Frost (1986). Skin color preference, sexual dimorphism and sexual selection: A case of gene-culture co-evolution? Ethnic and Racial Studies, 9, 87-113.
Wang, D.Y., T.J.A. Key, M.C. Pike, J. Boreham, and J. Chen (1991). Serum hormone levels in British and rural Chinese females. Breast Cancer Research and Treatment, 18, S41-S45.
* * *
Source: Author






If I were Mr. Spock’s “Star Trek” I would say, “fascinating.”
However, since I am not, I will not.
I don’t know why but I find this sort of thing interesting. I know Europeans are fairly unique in their variation, but I am also curious as to how it relates to the East Asians, as they seem the closest to us genetically when compared with any non-White race.